NCERT LINE BY LINE QUESTIONS
- Work function depends on [NCERT Pg. 387]
(1) Metal only (2) Nature of surface only
(3) Both metal and nature of surface (4) Threshold frequency
2. Saturation photoelectric current [NCERT Pg. 391]
(1) Increase with increase in plate potential
(2) Increase with decrease in plate in plate potential
(3) Is independent of plate potential
(4) Increase with increase in frequency
3. Monochromatic light of frequency 6 X 1014 Hz is produced by a laser. The power emitted is
2 X 10–3W. The number photons emitted per second by source is [NCERT Pg. 396]
(1) 5.0 X 1015 (2) 5.0 X 1016 (3) 5.0 X 1017 (4) 5.0 X 1018
4. A particle is moving three times as fast as an electron. The ratio of de-Broglie wavelength of particle to that of electron is 1.813 X 10–4. The particle may be [NCERT Pg. 402]
(1) Proton (2) Deutron (3) a-particle (4) Triton
5. An electron microscope uses electrons accelerated by a voltage of 50 kV. how does the resolving power of this electron microscope compare with that of an optical microscope which uses yellow light? [NCERT Pg. 411]
(1) 104 times (2) 105 times (3) 106 times (4) 103 times
6. A particle is dropped from a height H. The de-Broglie wavelength of the particle as a function of height is proportional to [NCERT Pg. 400]
(1) H (2) H1/2 (3) H° (4) H–1/2
7. A proton and an a-particle are accelerated through the same potential difference. The ratio of de-Broglie wavelength to that is [NCERT Pg. 400]
(1) (2) 2 : 1 (3) (4)
8. Which of the following statements is incorrect about the photons?
[NCERT Pg. 396]
(1) Momentum of photon is (2) Rest mass of photon is zero
(3) Photons exert no pressure (4) Energy of photon is frv
9. The wavelength of matter wave is independent of [NCERT Pg. 398]
(1) Mass (2) Velocity (3) Kinetic energy (4) Charge
10. Which experiment best support the theory that matter has wave nature? [NCERT Pg. 403]
(1) Photoelectric effect (2) -scattering experiment
(3) Davisson and Germer experiement (4) Compton effect
11. Which among the following phenomenon shows particle nature of light? [NCERT Pg. 395]
(1) Photoelectric effect (2) Interference
(3) Polarization (4) Matter waves
12. Which of the following device is some times called electric eye? [NCERT-Pg399]
(1) Light emitting diode (2) Photocell
(3) Electric generator (4) Integrated chip
13. For a certain metal, incident frequency v is five times of threshold frequency vo and maximum speed of coming out photoelectrons is 8 X 106 m/s. If v = 2vo, the maximum speed of photoelectrons will be [NCERT Pg. 395]
(1) 4 x 106 m/s (2) 6 x 106 m/s (3) 3 x 106 m/s (4) 1 x 106 m/s
14, An electron is moving with an initial velocity enters in a uniform magnetic field
. Then its de-Broglie wavelength [NCERT Pg. 400]
(1) Increase with time (2) Decrease with time
(3) Remains constant (4) Increases and decreases periodically
15. For a wavelength of 400 nm, kinetic energy of emitted photoelectron is twice that for a wavelength of 600 nm from a given metal. The work function of metal is [NCERT Pg. 395]
(1) 1.03 eV (2) 2 11 eV (3)4.14eV (4) 2.43 eV
16. The linear momentum of a 3 MeV photon is [NCERT Pg. 398)
(1) 0.01 eV s m–1 (2) 0.02 eV s m–1
(3) 0.03 eV s m–1 (4) 0.04 eV s m–1
17. A particle of mass 4m at rest decays into two particles of mass m and 3m. The ratio of de-
Broglie wavelength of two particles will be [NCERT Pg. 400)
(1) (2) 4 (3) 2 (4) 1
18. In a photon particle collision (such as photon electron collision). Which of the following may not be conserved? (NCERT Pg. 396]
(1) Total energy (2) Number of photons
(3) Total momentum (4) None of above
19. If the momentum of an electron is changed by P, then the de-Broglie wavelength associated with it changes by 0.5%. The initial momentum of electron will be [NCERT Pg 400]
(1) 200P (2) 400P (3) (4) 100P
20. The phenomena of photoelectric effect was first explained by [NCERT Pg. 395]
(1) Albert Einstein (2) Heinrich Hertz
(3) Wilhelm Hallwachs (4) Philipp Lenard
NCERT BASED PRACTICE QUESTIONS
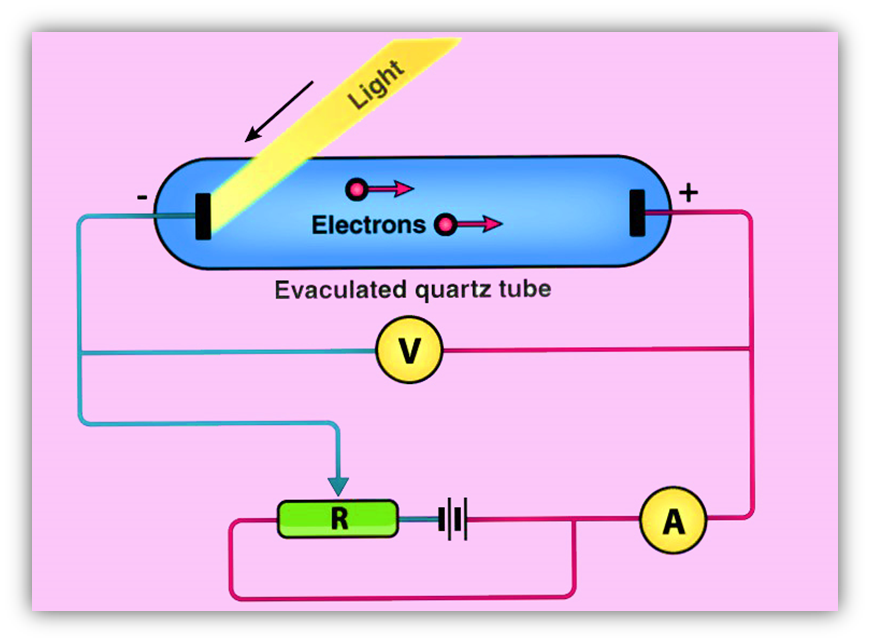
- The Photoelectric current is directly proportional to
(a) Intensity of incident light (b) Frequency
(c) Both (a) and (b) (d) None of these
2. The maximum kinetic energy of the photoelectrons depends upon
(a) Intensity of Incident light (b) Frequency
(c) Both (a) and (b) (d) None of these
3. The momentum of a photon of energy I MeV in kg ms-1 will be
(a) 10-22 (b) 0.33 x 106 (c) 5 x 10-22 (d) 7 x 10-24
4. The emission of electrons is possible by
(a) Photoelectric effect (d) thermionic effect
(c) Both (a) & (b) (d) None of these
5. Which of the following has highest specific charge?
(a) Positron (b) Proton (c) Helium (d) None of these
6. The magnitude of saturation photoelectric current depends upon
(a) frequency (b) Intensity (c) Work function (d) Stopping potential
7. The thermions are
(a) photons (b) Positron (c) Proton (d) Electrons
8. The rest mass of photon is
(a) 1.76 x 10-35Kg (b) Zero (c) 9 x 10-31Kg (d) 1a.m.u
9. Energy of the photon cannot be represented by
(a) hv (b) hc (c) hc/ (d) hcv
10. The momentum of a photon is p . the corresponding wavelength is
(a) h/p (b) hp (c) p/h (d)
11. Velocity of photon is proportional to
(a) (b) (c) V (d) V2
12. Monochromatic light of frequency 6.0 X 1014 Hz is produced by a laser. The power emitted is 2.0 x 10-3 W. What is the energy of a photon the light beam?
(a) 398 x 10-19 J (b) 3.98 x 10-19 J (c) 3.8 x 10-19 J (d) None
13. The work function of cesium is 2.14 eV. Find the threshold frequency for cesium.
(a) 5.16 x 1014 Hz (b) 51.6 x 1014 Hz (c) 5.16 x 1010 Hz (d) None
14. What is the de Broglie wavelength associated with an electron moving with a speed of 5.4 m/s ?
(a) 0.135 mm (b) 0.135 nm (c)135 mm (d) None
15. An electron, an particle, and a proton have the same kinetic energy. Which of these particles has the shortest de Broglie wavelength?
(a) -particle (b) Both (c) proton (d) None
16. A particle is moving three times as fast as an electron. The ratio of the de Broglie wavelength of the particle to that of the electron is 1.813. Calculate the particle’s mass.
(a) 1.67510-27 kg (b) 1.67510-26 kg (c) 16.7510-27 kg (d) None
17. What is the de Broglie wavelength associated with an electron, accelerated through a potential difference of 100 volts.
(a) 0.125nm (b) 0.123nm (c) 0.130nm (d) None
18. The work function of cesium metal is 2.14 eV. When light of frequency 6is incident on the metal surface, photo emission of electrons occurs. What is the maximum kinetic energy of the emitted electrons?
(a) 0.54 x 10-19 J (b) 54 x 10-19 J (c) 0.054 (d) 54.0 x10-19 J
19. The photoelectric cut – off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
(a) 2.4 x 10-19 J (b) 14 x 10-19 J (c) 3.4 x 10-19 J (d) None
20. The energy flux of sunlight reaching the surface of the earth is 1.388 How many photons (nearly) per square meter are incident on the Earth per second? Assume that the photons in the sunlight have an average wavelength of 550nm.
(a) 4 x 1021 photons/m2 s (b) 4 x1022 photons/m2 s
(c) 4 x 1023 photons/m2 s (d) 4 x 1027 photons/m2
21. In an experiment on photoelectric effect , the slope of the cut – off voltage versus frequency of incident light is found to be 4.12 Vs. Calculate the value of planck’s constant.
(a) 65.9 x 10-34 Js (b) 6.59 x 10-34 Js (c) 6.9 x 10-34 Js (d) None
22. A 100W sodium lamp radiates energy uniformly in all directions. The lamp is located at the centre of a large sphere that absorbs all the sodium light which is incident on it. The wavelength of the sodium light is 589 nm.
What is the energy per photon associated with the sodium light?
(a) 3.38×10-19 J (b) 38×10-19 J (c) 3.38×10-34 J (d) None
23. The threshold frequency for a certain metal is 3.3 Hz. If light of frequency 8.2 is incident on the metal, predict the cut – off voltage for the photoelectric emission.
(a) 20 V (b) 200 V (c) 2.0 V (d) None
24. The work function for a certain metal is 4.2 eV. Will this metal give photoelectric emission for incident radiation of wavelength 330 nm?
(a) Yes (b) No (c) may or may not (d) None of these
25. Light of frequency 7.21 is incident on a metal surface. Electrons with a maximum speed of 6.0 m/s are ejected from the surface. What is the threshold frequency for photoemission of electrons?
(a) 4.73×1014 Hz (b) 47.3×1014 Hz (c) 47×1014 Hz (d) None
26. Light of wavelength 488 nm is produced by an argon laser which is used in the photoelectric effect. When light from this spectral line is incident on the emitter, the stopping (cut – off) potential of photoelectrons is 0.38 V. Find the work function of the material from which the emitter is made.
(a) 3.46 x 10-19 J (b) 34.6 x 10-19 J (c) 346 x 10-19 J (d) None
27. Calculate the momentum of the electrons accelerated through a potential difference of 56 V.
(a) 40×10-24 kg ms-1 (b) 4.04×10-24 kg ms-1
(c) 4×10-24 kg ms-1 (d) None
28 What is the momentum of an electron with kinetic energy of 120 eV.
(a) 5.92 x 10-24 kg ms-1 (b) 59.2 x 10-24 kg ms-1
(c) 5.92 x 10-28 kg ms-1 (d) None
29 What is the de Broglie wavelength of a bullet of mass 0.040 kg traveling at the speed of 1.0 km/sec.
(a) 17 x 10-35 m (b) 1.7 x 10-35 m (c) 19 x 10-35 m (d) None
30. The wavelength of light form the spectral emission line of sodium is 589 nm, Find the kinetic energy.
(a) 4.34 eV (b) 43.4 eV (c) 434 eV (d) None
31. An electron of wavelength of 1.00 nm. Find the moment of electron.
(a) 6.63 kg m/s (b) .663 kg m/s (c) 66 kg m/s (d) None
32. For what kinetic energy of a neutron will the associated de Broglie wavelength be1.40
(a) 4.174 x 10-21eV (b) 4.174 x 10-2eV
(c) 4.174 x 10-34eV (d) None
33. What is the de Broglie wavelength of a nitrogen molecule in air at 300 K ? Assume that the molecule is moving with the root – mean – square speed of molecules at this temperature. (Atomic mass of nitrogen = 14.0076u)
(a) 28 nm (b) 0.28 nm (c) 0.028 nm (d) None
34 and gamma rays carry some momentum, which has the longest wavelength
(a) rays (b) rays
(c) gamma rays (d) all have same wavelength
35 If electron, proton and helium have same momentum, then de – Broglie’s wavelength decrease in order
(a) (b)
(c) (d)
36 Stopping potential required to reduce photoelectric current to zero
(a) Is directly proportional to the wavelength of incident radiation
(b) Is directly proportional to the frequency of the incident radiation
(c) Increases uniformly with wavelength of the incident radiation
(d) Decreases uniformly with the frequency of the incident radiation
1)a 2) b 3) c 4) c 5) a 6) b 7) d 8) b 9) c 10) a
11)b 12) b 13) a 14) b 15) a 16) a 17) b 18) a 19) a 20) a
21)b 22) a 23) c 24) b 25) a 26) a 27) b 28) a 29) b 30) a
31)a 32) b 33) c 34) d 35) d 36) b
